Report Description: Cardiac Marker Testing Market Size, Trend, by Product Type (Instruments & Devices, Reagents & Kits), by Biomarker Type, by Indication Type (Congestive Heart Failure, Myocardial Infarction, Acute coronary syndrome, Atherosclerosis, Ischemia), by End Users Type, by Region & Major Players – Global Forecast to 2030
Wissen Research analyzed that the global cardiac marker testing market was valued at USD 4.5 billion in 2024 and is projected to reach USD 7.6 billion by 2030, expected to grow at a CAGR of 9.2 % during the forecast period, 2025-2030.
Lucrative Opportunities: Cardiac Marker Testing Market
Global cardiac marker testing market is anticipated to reach USD 7.6 billion by 2030 from USD 4.5 billion in 2024, growing at an annualized rate of 9.2% during the forecast period, 2025-2030 | Europe is a key market player, supported by great healthcare systems and strong regulatory frameworks. Significant investment in cardiovascular research and a high prevalence of CVDs drive demand. These factors contribute to its substantial market share and influence. | Asia-Pacific The Asia-Pacific region is experiencing rapid market growth due to its large aging population and increasing CVD burden. Rising healthcare investments and improving infrastructure are key drivers. The expanding capabilities of local diagnostic companies further fuel this expansion. |
Technologies like High-Sensitivity Cardiac Troponin assays are enhancing the sensitivity of myocardial injury detection. Point-of-Care Testing (POCT) devices offer rapid results crucial for emergency diagnosis. Emerging molecular techniques explore improved quantification and new biomarker potential. | Research globally aims to validate novel biomarkers for better risk stratification in heart failure and other conditions. Integration of biomarker data with clinical/imaging info using AI is a major focus. Standardization and improving global accessibility of tests remain critical priorities. |
Strategic Activities
Drivers: Rising Prevalence of Cardiovascular Diseases
The primary driver behind the cardiac marker testing market is the increasing prevalence of cardiovascular diseases (CVDs) globally. This is confirmed by the World Health Organization (WHO), which states that CVDs are the leading cause of death worldwide, accounting for an estimated 17.9 million deaths per year. Cardiac marker tests—such as troponin, CK-MB, and myoglobin—are essential for early detection, diagnosis, and monitoring of acute coronary syndromes and other cardiac events in clinical practice. Their broad adoption is fundamentally supported by clinical guidelines and epidemiological trends, not by market speculation. For example, the European Society of Cardiology clinical guidelines recommend the use of cardiac troponin testing for rapid assessment in suspected myocardial infarction cases.
Opportunities: Expansion of Point-of-Care (POC) Testing and Technological Innovation.
Opportunity in the cardiac marker testing market arises from technological advances enabling point-of-care (POC) testing. Scholarly research demonstrates that POC cardiac marker assays allow for faster diagnosis and treatment, especially in emergency and outpatient settings. For instance, a 2022 study in the journal Clinical Chemistry found that high-sensitivity troponin assays used at the bedside can reduce diagnostic time for myocardial infarction by several hours and improve patient outcomes through earlier intervention. Regulatory agencies, including the US FDA, have approved several POC cardiac biomarker tests for use in emergency departments, cited in recent FDA device approvals.
Challenges: High Costs and Accessibility Barriers in Cardiac Marker Testing
A substantial challenge constraining the cardiac marker testing market is the significant cost associated with advanced diagnostic equipment and high-sensitivity assays, which creates barriers to widespread accessibility, particularly in low- and middle-income countries. This financial hurdle presents a multi-layered difficulty impacting both healthcare providers and patients globally. Healthcare facilities face considerable capital investment requirements for sophisticated cardiac testing platforms, including automated immunoassay analyzers capable of high-sensitivity cardiac troponin measurements and Point-of-Care Testing (POCT) devices, alongside the ongoing costs for specialized reagents, maintenance, staff training, and necessary infrastructure. Research indicates that the expense of advanced cardiac biomarker panels and high-sensitivity assays can strain healthcare budgets, especially in resource-limited settings. Reimbursement challenges further compound the cost issue, as inconsistent or insufficient coverage for certain cardiac tests can limit their adoption and availability, while patients in many regions face out-of-pocket expenses for these critical diagnostics, creating significant financial barriers that can delay or prevent timely assessment and treatment for cardiovascular conditions.
The global cardiac marker testing market is a vital segment within healthcare, essential for the rapid and accurate diagnosis, risk stratification, and monitoring of cardiovascular diseases that remain a leading cause of mortality globally. Valued at approximately USD 4.5 billion in 2024, this market is anticipated to experience steady growth, projected to exceed USD 7.6 billion by 2030, reflecting a Compound Annual Growth Rate (CAGR) of about 9.2%. This expansion is primarily driven by the increasing global burden of cardiovascular diseases (CVDs), including heart attacks and heart failure, fueled by aging populations, rising prevalence of risk factors like obesity and diabetes, and improved access to healthcare services requiring diagnostic interventions. Significant opportunities arise from technological advancements such as high-sensitivity cardiac troponin assays enabling earlier detection, the growing adoption of point-of-care (POC) testing devices for faster results in acute settings, and the integration of artificial intelligence (AI) to enhance data interpretation and risk prediction, collectively improving patient outcomes through timely and precise cardiac assessment.
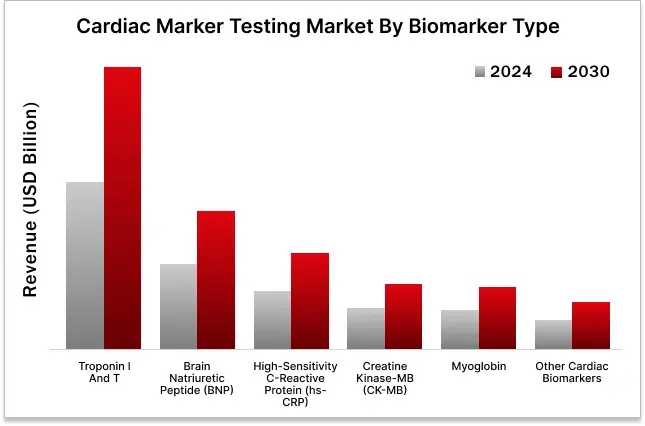
In the cardiac marker testing market, Troponin I and T held the largest share by biomarker type in 2024.
Diagnostic Testing Facilities held the largest share in the cardiac marker testing market by end user in 2024.

Asia Pacific will show the highest growth rate in the cardiac marker testing market in the forecast period. (2025-2030)
This rapid expansion is fueled by a number of factors, including the escalating burden of cardiovascular diseases (CVDs) driven by aging populations, increasing urbanization, sedentary lifestyles, and rising rates of diabetes and obesity across major economies like China and India. Governments in the region are significantly investing in healthcare infrastructure modernization and improving access to diagnostic services, often coupled with policy reforms expanding insurance coverage for cardiovascular care. There is a growing awareness among both the public and clinicians regarding the critical role of rapid and accurate cardiac marker testing in diagnosing conditions like myocardial infarction and heart failure, particularly in emergency settings. The region is also rapidly adopting advanced diagnostic technologies, including high-sensitivity cardiac troponin assays and point-of-care testing (POCT) devices, which enhance diagnostic speed and accessibility. Furthermore, Asia-Pacific’s large population base and increasing participation in global clinical trials make it an attractive destination for international diagnostics and pharmaceutical companies seeking to develop, validate, and launch new cardiac testing solutions. Strong government initiatives focused on improving public health outcomes, strengthening national healthcare systems, and adopting international diagnostic standards are pivotal. Support from international organizations and enhanced regional collaborations are also bolstering diagnostic capacity. These dynamics are stimulating substantial investments, the development of local manufacturing capabilities, and the introduction of innovative cardiac marker testing solutions tailored to the region’s specific needs, driving significant market growth in Asia-Pacific.
BRAND ANALYSIS: CARDIAC MARKERS
“Multiplexed proteomic and metabolomic platforms are allowing for the simultaneous measurement of a broader panel of cardiac and inflammation-related biomarkers, moving beyond single-marker approaches to provide a more holistic assessment of myocardial injury, hemodynamic stress, and overall cardiovascular risk profile.”
Professor of Cardiovascular Medicine – University Hospital Research Center (North America)
“The integration of high-sensitivity cardiac troponin assays with advanced algorithms for kinetic modeling and patient clinical data is enabling more precise discrimination between true myocardial infarction and non-AMI causes of chest pain, facilitating rapid risk stratification and safe early discharge in emergency settings.”
Lead Clinician – Academic Emergency Department, Cardiovascular Research Division (Europe)
“Point-of-care testing (POCT) devices featuring rapid, automated cardiac marker assays are significantly reducing time-to-results in critical care and emergency environments, directly correlating with faster clinical decision-making and improved patient outcomes in acute coronary syndrome management.”
Head of Clinical Chemistry & POCT – Large Tertiary Care Hospital Network (Asia-Pacific)
“The application of artificial intelligence and machine learning to longitudinal patterns of cardiac biomarker changes, alongside imaging and ECG data, holds promise for predicting disease progression, identifying individuals at high risk of adverse events, and personalizing therapeutic interventions beyond acute diagnosis.”
Director of Translational Cardiology – Biotechnology Research Institute (North America)
Sources: Primary Research and Wissen Research Analysis.
Note: Above mention is non-exhaustive samples of the primary insights.
1. INTRODUCTION
1.1. KEY OBJECTIVES
1.2. DEFINITIONS
1.2.1. IN SCOPE
1.2.2. OUT OF SCOPE
1.3. SCOPE OF THE REPORT
1.4. SCOPE RELATED LIMITATIONS
1.5. KEY STAKEHOLDERS
2. RESEARCH METHODOLOGY
2.1. RESEARCH APPROACH
2.2. RESEARCH METHODOLOGY / DESIGN
2.3. MARKET SIZE ESTIMATION APPROACHES
2.3.1. SECONDARY RESEARCH
2.3.2. PRIMARY RESEARCH
2.3.2.1. KEY INSIGHTS FROM INDUSTRY EXPERTS
2.4. DATA VALIDATION & TRIANGULATION
2.5. ASSUMPTIONS OF THE STUDY
3. EXECUTIVE SUMMARY & PREMIUM CONTENT
3.1. GLOBAL MARKET OUTLOOK
3.2. KEY MARKET FINDINGS
4. MARKET OVERVIEW
4.1. CARDIAC MARKER TESTING MARKET: OVERVIEW
4.1.1. INTRODUCTION
4.2. MARKET DYNAMICS
4.2.1. MARKET DRIVERS
4.2.2. MARKET OPPORTUNITIES
4.2.3. RESTRAINTS/CHALLENGES
4.3. END USER PERCEPTION
4.4. REGULATORY SCENARIO & TRENDS
4.5. NEED GAP ANALYSIS
4.6. SUPPLY CHAIN / VALUE CHAIN ANALYSIS
4.7. CASE STUDY ANALYSIS
4.8. TECHNOLOGY ANALYSIS
4.9. TRADE ANALYSIS
4.10. INDUSTRY TRENDS
4.11. PRICING ANALYSIS
4.12. REIMBURSEMENT SCENARIO
4.13. USE OF AI IN CARDIAC MARKER TESTING DEVICE
4.14. PORTER’S FIVE FORCES ANALYSIS
4.15. REGULATORY LANDSCAPE
4.15.1. NORTH AMERICA
4.15.2. EUROPE
4.15.3. ASIA PACIFIC
5. PATENT ANALYSIS
5.1. TOP ASSIGNEES
5.2. GEOGRAPHY FOCUS OF TOP ASSIGNEES
5.3. LEGAL STATUS
5.4. TECHNOLOGY EVOLUTION
5.5. KEY PATENTS
5.6. PATENT TRENDS AND INNOVATIONS
6. GLOBAL CARDIAC MARKER TESTING MARKET, BY PRODUCT TYPE (2024-2030, USD MILLION)
6.1. INSTRUMENTS & DEVICES
6.1.1.ELISA
6.1.2.IMMUNOCHROMATOGRAPHY
6.1.3.IMMUNOFLUORESCENCE
6.1.4.CHEMILUMINESCENCE
6.2. REAGENTS & KITS
7. GLOBAL CARDIAC MARKER TESTING MARKET, BY BIOMARKER TYPE (2024-2030, USD MILLION)
7.1. CREATINE KINASE-MB
7.2. BRAIN NATRIURETIC PEPTIDE
7.3. MYOGLOBIN
7.4. HIGH SENSITIVITY C-REACTIVE PROTEIN
7.5. TROPONIN I AND T
7.6. OTHER CARDIAC BIOMARKER
8. GLOBAL CARDIAC MARKER TESTING MARKET, BY INDICATION TYPE (2024-2030, USD MILLION)
8.1. CONGESTIVE HEART FAILURE
8.2. MYOCARDIAL INFARCTION
8.3. ACUTE CORONARY SYNDROME
8.4. ATHEROSCLEROSIS
8.5. ISCHEMIA
9. GLOBAL CARDIAC MARKER TESTING MARKET, BY END USERS TYPE (2024-2030, USD MILLION)
9.1. DIAGNOSTIC TESTING FACILITIES
9.1.1.HOSPITAL LABORATORIES
9.1.2.CONTRACT TESTING LABORATORIES
9.2. POINT OF CARE TESTING FACILITIES
9.3. ACADEMIC & RESEARCH INSTITUTIONS
10. GLOBAL CARDIAC MARKER TESTING MARKET, BY REGION (2024-2030, USD MILLION)
10.1. NORTH AMERICA
10.1.1. US
10.1.2. CANADA
10.2. EUROPE
10.2.1. GERMANY
10.2.2. FRANCE
10.2.3. SPAIN
10.2.4. ITALY
10.2.5. UK
10.2.6. REST OF THE EUROPE
10.3. ASIA-PACIFIC
10.3.1. CHINA
10.3.2. JAPAN
10.3.3. INDIA
10.3.4. AUSTRALIA AND NEW ZEALAND
10.3.5. SOUTH KOREA
10.3.6. REST OF THE ASIA-PACIFIC
10.4. MIDDLE EAST AND AFRICA
10.5. LATIN AMERICA
11. COMPETITIVE ANALYSIS
11.1. PRODUCT PIPELINE: CARDIAC MARKER TESTING DEVICES
11.2. KEY PLAYERS FOOTPRINT ANALYSIS
11.3. MARKET SHARE ANALYSIS (2023/2024)
11.4. REGIONAL SNAPSHOT OF KEY PLAYERS
11.5. R&D EXPENDITURE OF KEY PLAYERS
12. COMPANY PROFILES
12.1. F. HOFFMANN-LA ROCHE LTD. (SWITZERLAND)
12.1.1. BUSINESS OVERVIEW
12.1.2. PRODUCT PORTFOLIO
12.1.3. FINANCIAL SNAPSHOT
12.1.4. RECENT DEVELOPMENTS
12.1.4.1. MERGER/ACQUISITIONS
12.1.4.2. PRODUCT APPROVAL/LAUNCHES
12.1.4.3. PARTNERSHIP/COLLABORATIONS/AGREEMENTS
12.2. BECTON, DICKINSON AND COMPANY (BD) [US]
12.3. ABBOTT LABORATORIES [US]
12.4. THERMO FISHER SCIENTIFIC INC. [US]
12.5. BIO-RAD LABORATORIES, INC. [US]
12.6. BIOMÉRIEUX [FRANCE]
12.7. SIEMENS HEALTHINEERS AG [GERMANY]
12.9. QUIDEL CORPORATION [US]
12.10. PERKINELMER INC. [US}
12.11. TOSOH CORPORATION [JAPAN]
12.12. DANAHER CORPORATION [US]
12.13. LSI MEDIENCE CORPORATION [JAPAN]
12.14. OTHER PLAYERS
12.14.1. RANDOX LABORATORIES [UK]
12.14.2. GUANGZHOU WONDFO BIOTECH CO., LTD [CHINA]
12.14.3. BODITECH MED INC. [SOUTH KOREA]
12.14.4. TULIP DIAGNOSTIC (P) LTD. [INDIA]
12.14.5. BTNX, INC. [CANADA]
12.14.6. RESPONSE BIOMEDICAL [CANADA]
12.14.7. ALFA SCIENTIFIC DESIGN, INC. [US]
12.14.8. CTK BIOTECH, INC. [US]
12.14.9. CREATIVE DIAGNOSTICS [US]
12.14.10. LIFESIGN LLC. [US]
12.14.11. CARDIOGENICS HOLDING INC. [CANADA]
12.14.12. ATLAS MEDICAL GMBH [GERMANY]
13. APPENDIX
13.1. INDUSTRY SPEAK
13.2. QUESTIONNAIRE/DISCUSSION GUIDE
13.3. AVAILABLE CUSTOM WORK
13.4. ADJACENT STUDIES
13.5. AUTHORS.
14. REFERENCES
Market Definition: Cardiac Marker Testing
The Cardiac Marker Testing Market is a significant and fast-growing market in healthcare. It focuses on analysing certain biochemical markers in the blood to diagnose, assess risk, and monitor heart diseases, especially acute coronary syndromes like heart attacks. This market includes various products such as assay kits, reagents, and advanced analyzers, both for central laboratories and point-of-care devices. These products detect markers like Troponins, CK-MB, and BNP. By offering quick and accurate information about heart injury or stress, these tests help differentiate between cardiac and non-cardiac conditions early on. They support risk assessment, inform crucial treatment choices, and assist in tracking disease progression and treatment response. This capability plays a crucial role in improving patient outcomes and managing both cardiovascular emergencies and chronic illnesses effectively.
FIGURE: CARDIAC MARKER TESTING MARKET SEGMENTS
Sources: Company Websites and Wissen Research Analysis
Key Stakeholders
Key Study Objectives
Research Methodology
The objective of the study is to analyze the key market dynamics such as drivers, opportunities, challenges, restraints, and key player strategies. To track company developments such as product launches and approvals, expansions, and collaborations of the leading players, the competitive landscape of the cardiac marker testing market to analyze market players on various parameters within the broad categories of business and product strategy. Top-down and bottom-up approaches will be used to estimate the market size. To estimate the market size of segments and sub segments the market breakdown and data triangulation will be used.
FIGURE: RESEARCH DESIGN
Sources: Wissen Research Analysis
Research Approach
Collecting Secondary Data
The secondary research data collection process involves the usage of secondary sources, directories, databases, annual reports, investor presentations, and SEC filings of companies. Secondary research will be used to identify and collect information useful for the extensive, technical, market-oriented, and commercial study of the cardiac marker testing market. A database of the key industry leaders will also be prepared using secondary research.
Collecting Primary Data
The primary research data will be conducted after acquiring knowledge about the cardiac marker testing market scenario through secondary research. A significant number of primary interviews will be conducted with stakeholders from both the demand side and supply side (including various industry experts, such as Directors, Chief X Officers (CXOs), Vice Presidents (VPs) from business development, marketing and product development teams, product manufacturers) across major countries of North America, Europe, Asia Pacific, and Rest of the World. Primary data for this report was collected through questionnaires, emails, and telephonic interviews.
Market Size Estimation
All major developers offering various cardiac marker testing will be identified at the global/regional level. Revenue mapping will be done for the major players, which will further be extrapolated to arrive at the global market value of each type of segment. The market value of cardiac marker testing market will also split into various segments and sub segments at the region level based on:
Research Design
After arriving at the overall market size-using the market size estimation processes-the market will be split into several segments and sub segment. To complete the overall market engineering process and arrive at the exact statistics of each market segment and sub segment, the data triangulation, and market breakdown procedures will be employed, wherever applicable. The data will be triangulated by studying various factors and trends from both the demand and supply sides in the cardiac marker testing industry.
The global market for cardiac marker testing market was valued at USD 4.5 billion in 2024.
The global cardiac marker testing market is anticipated to grow at an annual growth rate of 9.2% from 2025 to 2030 to reach USD 7.6 billion, by 2030.
Leading players within the cardiac marker testing market are Abbott Laboratories [US], bioMérieux [France], Siemens Healthineers AG [Germany], F. Hoffmann-La Roche Ltd. (Switzerland), Danaher Corporation [US]
The cardiac marker testing market is characterized as moderately consolidated, dominated by a few large multinational companies that hold significant market share. These major players typically have broad portfolios encompassing a wide range of in vitro diagnostics, including advanced cardiac biomarker assays (like Troponin, BNP/NT-proBNP) and the sophisticated analyzers required for their detection, often integrating them into comprehensive laboratory information systems.
