
Close


Overview:
About RT
Nuclear medicine, born from the discovery of radioactivity in the early 20th century, evolved with key milestones like technetium-99m in the 1950s. SPECT and PET scans later revolutionized diagnostics. Ongoing research ensures nuclear medicine remains at the forefront of medical advancements, blending history with cutting-edge technology.
Nuclear medicine, a pioneering medical specialty, harnesses the power of radioactive tracers, or radiopharmaceuticals, to delve into the intricacies of bodily functions, diagnose diseases, and advance treatment modalities. The cornerstone of this field lies in cutting-edge imaging technologies, notably Single Photon Emission Computed Tomography (SPECT) and Positron Emission Tomography (PET) scans.
Radioactive tracers, the linchpin of nuclear medicine, consist of carrier molecules tightly bonded to a radioactive atom. These tracers, tailored for diverse diagnostic purposes, can interact with specific proteins, sugars, or even the patient’s cells. For instance, in pinpointing the source of intestinal bleeding, red blood cells labeled with radioactive atoms are injected, and their path is traced using SPECT scans, revealing crucial insights for effective intervention.
Administered via injection, inhalation, ingestion, or direct organ injection, radiopharmaceuticals undergo rigorous FDA scrutiny, meeting exacting standards for safety and efficacy. The choice of tracer dictates whether a SPECT or PET scan is employed, with each modality offering unique advantages.
SPECT imaging delivers three-dimensional tomographic images by detecting gamma ray emissions from injected tracers. The rotating gantry allows detectors to encircle the patient, capturing detailed insights into conditions like heart disease, bone disorders, gall bladder disease, and Parkinson’s disease.
In contrast, PET scans utilize radiotracers emitting positrons, which, upon interaction with electrons in the body, produce detectable photons. Predominantly employed for cancer detection and monitoring, PET scans excel in visualizing glucose utilization, crucial in identifying aggressive cancerous activity. PET/CT scanners, combining PET and CT scans, have become indispensable for comprehensive cancer staging.
Recently, the FDA approved a PET probe for Alzheimer’s disease diagnosis, offering a breakthrough in live imaging. This addresses the challenge of distinguishing Alzheimer’s from other forms of dementia while the patient is alive, revolutionizing diagnostic precision.
Beyond diagnostics, nuclear medicine plays a pivotal role in research, with the National Institute of Biomedical Imaging and Bioengineering (NIBIB) funding innovative endeavors to propel the field forward. These advancements promise not only refined diagnostic capabilities but also novel therapeutic approaches, marking nuclear medicine as a dynamic frontier in medical science.

Figure 1 Nuclear medicine
Credits: >>
For effective market positioning and assessing competitiveness over the short to mid-long term, a thorough understanding of the Radioactive Tracers development pipeline, including the evolving landscape, competitor initiatives, and expected advancements, is essential.
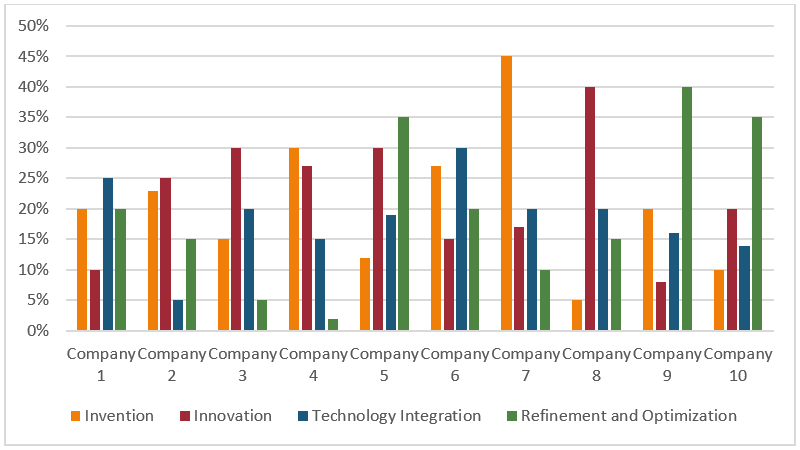
Figure 2 Ongoing technological Research
Profiles of stakeholders driving advancements in Radioactive Tracers solutions offer insights into financials, product portfolios, and recent innovations in the field.

Figure 3 Company Portfolio
This report examines patents related to Radioactive Tracers, uncovering key innovations and opportunities for improvements or collaborations in the field.
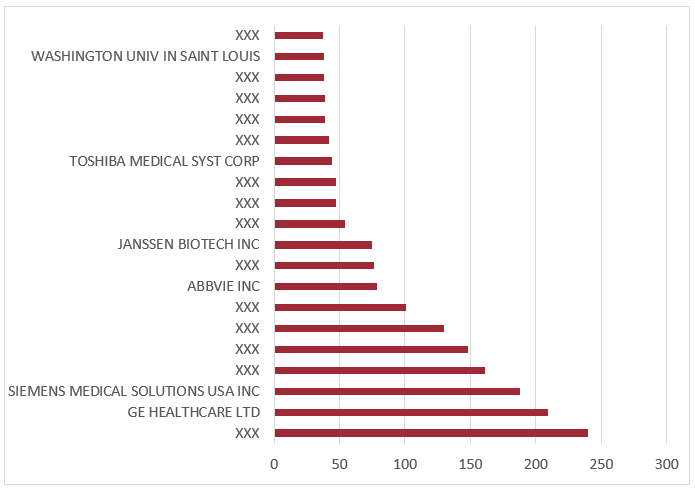
Figure 4 Top standard current assignees
This report on Radioactive Tracers anticipates emerging trends, growth opportunities, and pivotal factors shaping supply and demand dynamics concisely. It offers valuable quantitative insights into market size, revenue forecasts, and growth potentials in the field.
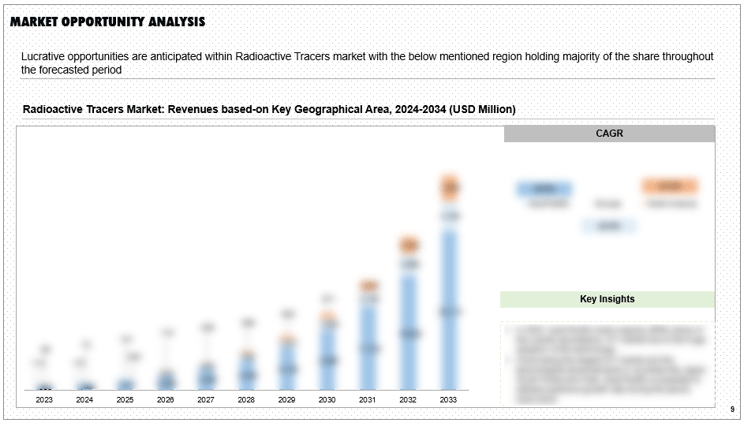
Figure 5 Market Opportunity Analysis
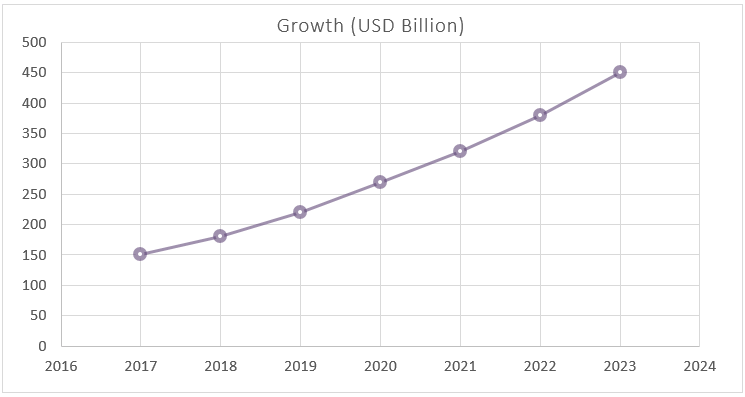
Figure 6 Yearly Market Growth, 2016-2024 (USD Billion)
(Segmented in terms of the financial growth)
This report on Radioactive Tracers spotlights prominent market players, offering concise profiles with information on market shares, strengths, weaknesses, and strategic approaches.
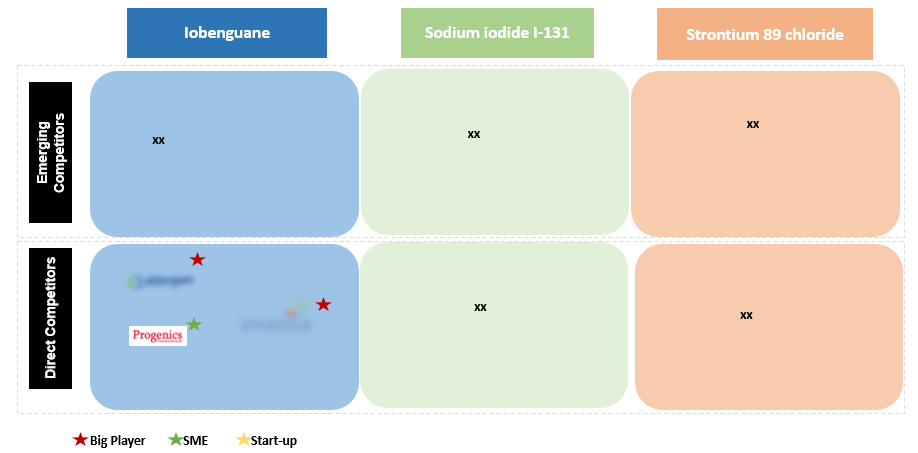
Figure 7 Navigating the Competitive part of Market landscaping
In order to give the most precise estimations and forecasts, Wissen Research uses an extensive and iterative research approach that is focused on reducing deviation. The company blends top-down and bottom-up methodologies for market segmentation and quantitative estimation. In addition, data triangulation, which examines the market from three separate angles, is a recurrent topic present in all of our research studies. Important components of the approach used for all of our studies include the following:
Preliminary data mining
On a wide scale, unprocessed market data is collected. Continuous data filtering makes sure that only verified and authenticated sources are taken into account. Additionally, data is extracted from a wide range of reports in our repository and from a number of reputable premium databases. We gather information from raw material suppliers, distributors, and purchasers to help with this since understanding the entire value chain is crucial for a thorough understanding of the market.
Surveys, technical symposia, and trade magazines are used to gather information on technical concerns and trends. Technical information focusing on white space and freedom of movement is also obtained from an intellectual property standpoint. Additionally, information on the industry’s drivers, constraints, and pricing patterns is obtained. As a result, a variety of original data are included in the material that is then cross-validated and certified with published sources.
Statistical model
We use simulation models to generate our market projections and estimates. Every study receives a special model that is tailored to it. Data for market dynamics, the technology environment, application development, and pricing patterns are gathered and supplied into the model all at once for analysis. The relative relevance of these factors is investigated, and their impact on the forecast period is assessed, using correlation, regression, and time series analysis. The process of market forecasting combines technological analysis with economic strategies, practical business acumen, and subject expertise.
Econometric models are frequently used for short-term forecasting, but technology market models are typically employed for long-term forecasting. These are based on a confluence of the business environment, regulatory environment, economic projection, and technical landscape. In order to develop global estimates, it is preferable to estimate markets from the bottom up by integrating data from key regional markets. This is required to ensure accuracy and a complete comprehension of the subject. Among the variables taken into account for forecasting are:
Regulations and anticipated developments
We give these criteria weights and use weighted average analysis to assess their market influence in order to calculate the anticipated market growth rate.
Primary research | Secondary research |
· Manufacturers · Technology distributors and wholesalers · End-user surveys · Consumer surveys | · Company reports and publications · Government publications · Independent investigations · Economic and demographic data · Online searches · Research reviews · Reference customers |
1. Introduction
1.1 Overview and Classification of Radioactive Tracers
1.2 Benefits and Applications
2. Overview of the Study
2.1 Objective
2.2 Technology categorization
2.3 Inclusion and Exclusion
2.4 Scope and Methodology
3. Executive Summary
3.1 Key Findings
3.2 Market Insights and Highlights
4. Patent Analysis
4.1 Overview of Radioactive Tracers Patents
4.2 Top Assignees
4.3 Geography Focus of Top Assignees
4.4 Legal Status
4.5 Assignee Segmentation
4.6 Network Analysis of Top Collaborating Entities
4.7 Technology Evolution
4.8 Key Patents
4.9 Patent Trends and Innovations
4.10 Key Players and Patent Portfolio Analysis
5. Clinical Trial Analysis
5.1 Overview of Clinical Trials
5.2 Analysis by Trial Registration Year
5.3 Analysis by Phase of Development
5.4 Analysis by Number of Patients Enrolled
5.5 Analysis by Status of Trial
5.6 Analysis by Study Design
5.7 Analysis by Type of Product
5.8 Analysis by Geography
5.9 Analysis by Key Sponsors/Collaborators
6. Product Analysis
6.1 Overview of Products in the Radioactive Tracers Market
6.2 Product Chapter
6.2.1 Approved
6.2.2 Pipeline
6.3 Emerging Products
7. Competitive Landscape
7.1 Benchmarking Parameters
7.2 Scoring of Different Companies
7.3 Top Players Profiles
7.3.1 Company X
7.3.2 Company Y
7.3.3 Company Z
7.4 Product Portfolio Analysis (Top Players)
7.5 Recent Developments and Strategies
8. Market Dynamics
8.1 Market Drivers and Challenges
8.2 Regulatory Landscape
8.3 Emerging Start-ups
8.4 Investment Trends
9. Market Forecast (2024-2033)
9.1 Methodology
9.2 Assumptions
9.3 Projected Growth of Radioactive Tracer Market (2024-2033)
9.3.1 Global Radioactive Tracer Market (2024-2033)
9.3.1.1 By Type
9.3.1.1.1 Therapeutic Radioactive Tracers
9.3.1.1.2 Diagnostic Radioactive Tracers
9.3.1.1.3 Others
9.3.1.2 By Application
9.3.1.2.1 Oncology
9.3.1.2.2 Cardiology
9.3.1.2.3 Others
9.3.2 Radioactive Tracers Market in USA and in UK & EU (4) Regions
9.3.2.1 By Type
9.3.2.1.1 Therapeutic Radioactive Tracers
9.3.2.1.2 Diagnostic Radioactive Tracers
9.3.2.1.3 Others
9.3.2.2 By Application
9.3.2.2.1 Oncology
9.3.2.2.2 Cardiology
9.3.2.2.3 Others
10. Future Outlook and Market Opportunities
10.1 Technological Advancements
10.2 Unmet Needs and Potential Market Gaps
10.3 Market Growth Opportunities
11. Conclusion
12. Appendix
12.1 Glossary of Terms
12.2 List of Abbreviations
13. References
S.no | Key Highlights of Report | |
1. | Patent Analysis | · Top Assignee · Geography focus of top Assignees · Assignee Segmentation · Network analysis of the top collaborating entities in Radioactive Tracers (RT) patent applications · Technology Evolution · Key Patents · Application and Issued Trend · Key technology |
2. | Market analysis | · Current Nuclear Medication Solutions and Applications · Emerging Technologies and Research Developments (by product analysis and scientific analysis) · Strategic activities · Operational Efficiency of the Nuclear Medication Solutions · Company portfolio · Detailed profiles of the key players that are engaged in the development of the Radioactive tracers. |
3. | Clinical Trials | · Analysis of clinical trial through graphical representation · Coverage of testing from the trial phases till commercialization |
4. | Forecast | · Detailed comprehension of the historic, current and forecasted trend of market by analysis of their impact on the market |
5. | Opportunity Analysis | · Technology evolution based on problem solution · Potential licensees · Advancement trends · Unmet needs · SWOT · Drivers and barriers |
6. | KOLs | · A detailed analysis and identification of the key opinion leaders (KOLs), shortlisted based on their contributions |
LIST OF FIGURES
Figure number | Description |
Figure 1 | Terminology of Radioactive Tracers (RT) Over The Years |
Figure 2 | Radioactive Tracers (RT)– History and Present |
Figure 3 | Projection of Radioactive Tracers (RT) till 2033 in different geographies |
Figure 4 | Technology Categorization For Radioactive Tracers (RT) |
Figure 5 | Recent Technology Trends in Radioactive Tracers (RT) |
Figure 6 | Technology Evolution in Radioactive Tracers (RT) |
Figure 7 | Geographical Distribution of Patents of Top Assignees |
Figure 8 | Assignee Segmentation (Companies) |
Figure 9 | Assignee Segmentation (Educational Establishment) |
Figure 10 | Patent Based Key Insights Of xx |
Figure 11 | Patent Based Key insights of xx |
Figure 12 | Patent Based Key insights of xx |
Figure 13 | Geographic Distribution of the Universities/Research Organizations Filling Patents On Various Technology advancements |
Figure 14 | Key Summary Regarding the Patent Filing On Radioactive Tracers (RT) |
Figure 15 | Product Pipeline of Different innovations with Companies Name |
Figure 16 | Portfolio for Government Approved Nuclear Medications |
Figure 17 | Key Growth Drivers for Radioactive Tracers (RT) Market |
Figure 18 | Restraints for Radioactive Tracers (RT) Market |
Figure 19 | xx Portfolio (Top Player) |
Figure 20 | xx Portfolio (Top Player) |
Figure 21 | xx Portfolio (Top Player) |
Figure 23 | xx Portfolio (Top Player) |
Figure 24 | xx Portfolio (Top Player) |
Figure 25 | xx Portfolio (Start-up) |
Figure 26 | xx Portfolio (Start-up) |
Figure 27 | xx Portfolio (Start-up) |
Figure 28 | Strategic Activities Including Collaboration, Partnerships and Acquisitions |
Figure 29 | Research Methodology for Patent, Selection and Analysis |
LIST OF GRAPHS
Graph number |
Description |
Graph 1 | Problem Solution Analysis |
Graph 2 | Top Assignees in Radioactive Tracers (RT) |
Graph 3 | Technology Focus of Top Assignees (IPC-CPC Classes) |
Graph 4 | Top Countries of Origin of Patents |
Graph 5 | New entrants in RT field |
Graph 7 | Legal Status |
Graph 8 | Most Cited Patents |
Graph 9 | Patents with Largest Invention Families |
Graph 10 | Most Claim-Heavy Patents |
Graph 11 | Filing Trends |
Graph 12 | Weighted Scores for Top 64 Players According to Benchmarking Criteria |
Graph 13 | Radioactive Tracers (RT) (CAGR: 2023-2033) |
Graph 14 | Radioactive Tracers (RT) Market Share: Distribution by Key Geographical Area, 2023-2033 |
LIST OF TABLES
Table number | Description |
Table 1 | Parameters included and excluded for conducting the analysis |
Table 2 | Technology Classes with Definitions |
Table 3 | Patent Litigation |
Table 4 | Highest Market Valued Patents |
Table 5 | SWOT Analysis of Top 3 Players |
Table 6 | Parameters and their score for Benchmarking |
Table 7 | Weighted scores for top 5 players according to benchmarking criteria |
Please Subscribe our news letter and get update.
© Copyright 2023 – Wissen Research All Rights Reserved.
Powered by VintageCoders